RJPS Vol No: 15 Issue No: 4 eISSN: pISSN:2249-2208
Dear Authors,
We invite you to watch this comprehensive video guide on the process of submitting your article online. This video will provide you with step-by-step instructions to ensure a smooth and successful submission.
Thank you for your attention and cooperation.
1Department of Pharmaceutics, R R College of Pharmacy, Chikkabanavara, Bangalore, Karnataka, India
2Dr. Hindustan Abdul Ahad, Professor & Head, Department of Pharmaceutics, R R College of Pharmacy, Chikkabanavara, Bangalore, Karnataka, India.
3Department of Quality Assurance, Himalaya Wellness Company, Tumkur Road, Makali, Bangalore, Karnataka, India
*Corresponding Author:
Dr. Hindustan Abdul Ahad, Professor & Head, Department of Pharmaceutics, R R College of Pharmacy, Chikkabanavara, Bangalore, Karnataka, India., Email: abdulhindustan@gmail.com
Abstract
Aim: The research aimed to investigate the enhanced mucoadhesive properties of Azadirachta indica fruit mucilage (AIFM) when incorporated into mucoadhesive microspheres containing Acyclovir as a representative model drug.
Methods: Nine formulations of mucoadhesive microspheres were created, incorporating Carbopol-934P and different ratios of AIFM. A central composite design was employed using Design-Expert software to assess the influence of independent variables (concentrations of polymers) on the dependent variable (mucoadhesive strength). The microspheres underwent compatibility studies and various physical evaluations, including analysis of Acyclovir content and its discharge kinetics.
Results: The study observed that the mucoadhesion strength of the microspheres increased as the levels of AIFM in the formulations increased. Additionally, formulations with lower levels of AIFM exhibited greater drug discharge at the 10th h mark. Conversely, higher AIFM concentrations in the formulations acted as retarding agents for drug discharge, leading to a slight reduction in the discharged amount of drug.
Conclusion: This experiment highlights that the mucoadhesive drug delivery system incorporating Acyclovir using Carbopol 934P along with AIFM successfully fulfills the requirement for gastro retention. This approach proves to be an effective strategy for delivering drugs specifically to the stomach.
Keywords
Downloads
-
1FullTextPDF
Article
Introduction
Emerging strategies are being pursued for drugdelivery to enhance the gastric availability of drugs while ensuring patient compliance. Gastro-retentive microspheres have gained significant attention due to their ease of preparation and administration.1 Acyclovir (AVR) is commonly used to treat viral infections. However, its oral bioavailability is limited (15-30%), with a short half-life of about 2 hrs. It is well absorbed in the stomach.2 Mucoadhesive systems are strongly reliant on the characteristics of the selected polymer. Many patients choose oral administration due to its convenience. While numerous polymers have been investigated for developing mucoadhesive compositions, these are frequently limited and expensive.3 In this context, researchers are seeking novel polymers from natural sources to enhance mucoadhesion. The study's focus is on investigating mucoadhesive microspheres using Azadirachta indica fruit mucilage (AIFM). In previous studies, AIFM has demonstrated antiviral activity, suggesting its potential for aiding antiviral therapy.4 This work, involving AVR-aided A. indica mucoadhesive microspheres (AAMM), aims to achieve sustained systemic drug availability over an extended period. Controlled discharge systems, designed for controlled and sustained drug discharge, offer a more intelligent approach for drugs with short half-lives or those requiring continuous dosing.
Materials and Methods
Materials
AVR was sourced from Ranbaxy Labs, Bangalore, while Carbopol 934P and dichloromethane were obtained from Merck, Hyderabad. All reagents used were of AR grade.
Methods
Extraction of mucilage
A. indica fruits were washed, peeled, soaked, and boiled for 1 hour. The mixture was left for another hour to release mucilage, after which seeds were removed and fractionated using petroleum ether (50%), ethyl acetate, butanol, and butanone. The mucilage was filtered through a muslin cloth, oven-dried at 40ºC, ground, sieved (#80), and stored in a desiccator at 30ºC with 45% relative humidity. As defined by Ahad et al. in 2021, this comprehensive procedure ensures the preparation and preservation of the extracted mucilage for further applications.5
Cleansing of the Mucilage
AIFM was homogenized (Biologics-150VT) with 5% trichloroacetic acid. The mixture was then centrifuged (Remi R-303), neutralized with NaOH, and dialyzed (SURDIAL-X) with distilled water to remove impurities. Mucilage was precipitated with 95% ethanol, collected, and purified through sequential washes with acetone and diethyl ether. This rigorous procedure culminated in the attainment of purified AIFM.
Experimental design
A central composite design (CCD) consisting of 9 runs was employed to create and analyze quadratic response surfaces for the optimization of AVR-A. indica mucoadhesive microspheres (AAMM) using Design-Expert software (version 11.0.5.0, Stat-Ease Inc.).6 The formulations were prepared using a central composite design, varying two key factors: Carbopol 934P (C934P) and AIFM, at three levels (-1: 50 mg, 0: 75 mg, +1: 100 mg) (Table 1). This systematic approach ensured precise optimization of polymer concentrations to achieve the desired mucoadhesive strength (MS) and controlled drug release for enhanced gastroretentive efficacy. The responses under consideration were the drug discharge at the 10th h (DR@10h) and the MS (g) for the AAMM.7,8
Preparation of AAMM
A mixture containing AVR, EC, Carbopol 934P, and AIFM was dissolved in acetic acid (2% v/v) and dichloromethane. Stirring was carried out using a three-bladed propeller stirrer (IKA-R1385) at 1500 rpm. This mixture was gradually added to liquid paraffin (which contained span 80). Additionally, glutaraldehyde was added dropwise at 1-hour intervals at a concentration of 4 minims per minute. The stirring persisted for three hours.9,10 After stirring, AAMMs were centrifuged, washed with petroleum ether to remove liquid paraffin, and treated with 5% sodium bisulfite for 15 minutes to remove glutaraldehyde. A final wash with distilled water was followed by drying and storage in vacuum desiccators.
Evaluation parameters
Determination of melting point
The melting point of AVR was determined by using the open capillary method.
Drug excipient compatibility studies
DSC
A 1:1 ratio of AVR and AIFM (10 mg) was placed in the mini pan of a DSC instrument and subjected to scanning within the temperature range of 50-300ºC (DSC 8000-Perkin Elmer).
Fourier transform infrared spectroscopy (FTIR)
The interaction between AVR and AIFM was examined using FTIR spectroscopy (Bruker), with scanning performed in the range of 4000-400 cm-1.
Initial risk assessment
The Quality Target Product Profile (QTPP), central to the QbD approach as per ICH Q8 and Q9, is established early in product development to ensure fundamental quality requirements. The QTPP and Critical Quality Attributes (CQAs) for the AAMM were determined using prior research and existing literature.
Evaluation of physical properties
Particle Size Measurement
The particle size of the AAMM was measured using a stage micrometer scale. Dry AAMM samples were placed on a clean glass slide and measured using an ocular micrometer. A minimum of 200 AAMM were counted for each batch.11
Production Yield
The production yield was calculated by dividing the average weight of dried AAMM (W1) recovered from each of the three trials by the initial dry weight (W2) (e.q.1).12
Entrapment Efficiency
AAMM samples weighing 100 mg were dispersed in 0.1 M HCl and subjected to overnight shaking. The resulting mixture was then filtered, and the filtrate was analyzed using a spectrophotometer (Elico Spectrophotometer, SL-174) at 254 nm.13 The entrapment efficiency (EE) was calculated by determining the ratio of the actual amount of the drug present in the formulation to the initial amount of the drug added (e.q.2).
Swelling Measurement
The swelling behavior of AAMM was investigated by immersing them in a solution of 0.1M HCl. After a period of 3 h, the AAMM samples were taken out, and subjected to centrifugation, and the increase in weight due to swelling was determined by calculating the difference between the weight at time t (Xt) and the initial weight at t = 0 (X0). This calculation was done according to the following equation (e.q.3).14
Where Xt-weight of the AAMM after time t; Xo- Initial weight of the AAMM.
Mucoadhesion Measurement Study
Mucoadhesive strength (MS) was measured using a sheep stomach obtained within 60 minutes of slaughter. The stomach was washed with isotonic saline, and AAMM was placed on its mucosal surface, which was fixed on a polyethylene plate at a 40º angle. A 0.1M HCl solution at 37±1ºC flowed over the tissue at 5 ml/min. The time for complete detachment of AAMM from the mucosal surface was recorded through visual inspection (e.q. 4).15
In vitro AVR Discharge Study
The in vitro discharge of AAMM was determined through a dissolution test conducted using the USP-II apparatus, maintaining a stirring rate of 50±5 rpm at a temperature of 37±0.5ºC. The dissolution medium used was 900 ml of 0.1N HCl. At various time intervals, a 5 ml sample was withdrawn from the medium, and the withdrawn volume was replaced with an equal volume of fresh dissolution medium.16 The collected samples were subsequently analyzed using spectrophotometry at a wavelength of 254 nm.
Statistical optimization
The Design-Expert software was utilized to assess the impact of independent variables on the responses, which were analyzed through contour plots (2D) and response surface plots (3D). The validity of polynomial models was statistically confirmed using ANOVA tables. A statistical model was developed based on the ANOVA results to determine model adequacy and significance. An F-value with a P-value of 0.05 or lower was considered statistically significant.17
Results
The freshly prepared A. indica fruit mucilage exhibited a yellowish-green color. It was utilized as an ingredient in the AAMM formulation, and its compatibility was evaluated.
Identification of drug
No significant alterations in the functional groups were observed when the AVR sample spectrum was compared with the reference spectrum.
Determination of Melting Point
The accepted melting point range for AVR was within 256.1±1.8ºC.
Compatibility studies
DSC analysis revealed pure AVR, with an endothermic peak at 256.19ºC, verifying purity. When coupled with excipients, the peak changed to 235.84ºC, showing interaction but not impacting AVR stability (Figure 1A). FTIR analysis of pure AVR showed peaks at 307.83 cm⁻¹ (secondary amine), 1040.88 cm⁻¹ (phenyl ester), and 1730.02 cm⁻¹ (carboxyl). The formulation blend (F-9) exhibited similar peaks at 3300.54 cm⁻¹, 1023.12 cm⁻¹, and 1727.11 cm⁻¹, confirming AVR's structural integrity (Figure 1B).
Quality Target Product Profile
The QTPP helped identify critical quality attributes (CQAs) and desired characteristics for AAMM. The preparation method was robust and reproducible, meeting quality standards. Using the QbD approach, CQAs like DR@10h and MS were analyzed, and the effects of AIFM and Carbopol 934P on AVR discharge and MS were thoroughly evaluated.
Physical properties
Formulations (F-1 to F-9) showed good flow and compressibility, with bulk density (0.327-0.596 g/cm³), tapped density (0.358-0.623 g/cm³), Carr’s index (3.565-9.515%), and Hausner ratio (<1.15). The angle of repose (19.58-25.58º) confirmed smooth flow, with F-7 and F-8 exhibiting optimal uniform blending and compression properties.
Particle size
Particle sizes of AAMM were determined using optical microscopy, revealing a range of 35.2±0.9 to 48.1±0.6 μm and F-5 exhibited a larger particle size.
Yield of AAMM
AAMM exhibited a % yield ranging from 82.2±1.2 to 96.2±2.3, with formulations F-4 and F-5 showing the highest yields.
% AVR entrapment
The EE of AVR in AAMM was ranged from 69.1±1.0 to 82.3±0.9, with F-6 and F-4 demonstrating the highest AVR entrapment values.
Swelling Measurement
The study highlights the relationship between mucoadhesion and AVR release in AAMM, revealing that lower AIFM concentrations reduced swelling indices. Batches F-1 to F-3 exhibited minimal swelling, while F-9 showed the highest. This suggests that insufficient AIFM in some batches hindered swelling, while higher AIFM levels in F-7 to F-9 enhanced it. The swelling behavior is attributed to AIFM's polar compounds, which aid water absorption and retention.
In vitro mucoadhesion
The mucoadhesion % of all batches of AAMM ranged from 17.3±0.12 g (F-1) to 19.3±0.05 g (F-9) (Figure 2A). It was observed that mucoadhesion increased with the higher content of AIFM.
In vitro drug discharge
The in vitro dissolution studies of AAMM formulations were conducted. Among the formulations, Formulations F-1 and F-2 exhibited the highest levels of AVR discharge, with 95.9±1.25% and 95.6±0.65%, respectively, at the end of the 10th h (Figure 2B).
AVR estimation
Using a UV-VIS spectrophotometer, the calibration curve for calculating AVR in a 0.1M HCl solution was established at the λmax of 254 nm. The Beer's law was observed to hold in the concentration range of 0-10 μg/ml. The calibration curve was constructed by three trials. This data is critical to ensure content homogeneity.
The discharge kinetics of AAMM were found to be best fitted to the Higuchi kinetics model. The dissolution results indicate that F-2 exhibited a superior AVR discharge profile to the other AAMMs.
Drug discharge kinetics
The responses obtained from these 9 formulations of AAMM were analyzed using Design-Expert 11.0 software. The results of the analysis of variance (ANOVA) revealed the model's F-value of 1202.70 confirms its significance, with only a 0.01% chance of occurring randomly. Model terms with P-values < 0.05, including A, B, and B², are significant, while values > 0.10 indicate insignificance.
Diagnostic analysis
DR@10h diagnostic analysis
Diagnostic plots (Figure 3A-D) assessed the adequacy of drug discharge at the 10th hour (DR@10h). The normal probability plot (Figure 3A) confirmed normality, with data points clustering around the normal line. The externally studentized residuals vs. predicted values plot (Figure 3B) indicated homoscedasticity, with points within limits. The residuals vs. run number plot (Figure 3C) showed consistency across experimental runs. The predicted vs. actual values plot (Figure 3D) demonstrated strong alignment, validating the model's accuracy and reliability.
Diagnostic analysis for MS
Diagnostic plots (Figure 3E–H) assessed the adequacy of MS and model fit. The normal probability plot (Figure 3E) confirmed normality, with residuals clustering around the probability line. The externally studentized residuals vs. predicted values plot (Figure 3F) indicated homoscedasticity. The residuals vs. run number plot (Figure 3G) showed consistency across runs. The predicted vs. actual values plot (Figure 3H) demonstrated strong alignment, confirming model accuracy.
ANOVA details of DR@10h
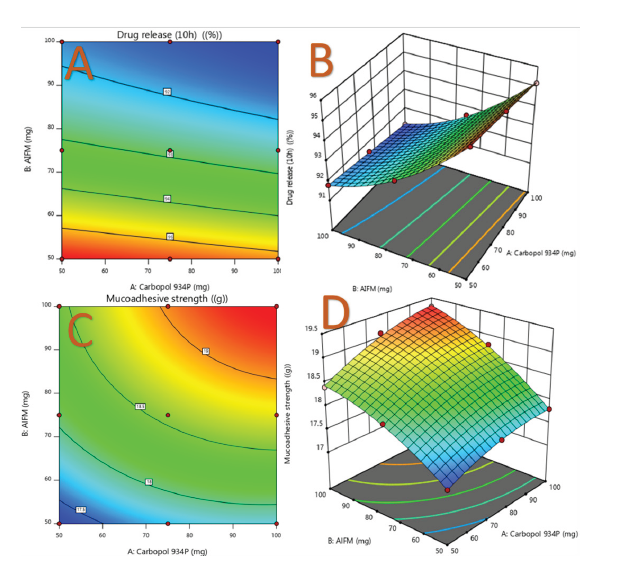
ANOVA results for DR@10h show a significant model with an F-value of 55.13, indicating only a 0.38% chance of occurring due to noise. Significant model terms (P < 0.05) include X1 and X2, while terms with P > 0.1 are insignificant. The coded equation for DR@10h is: DR@10h = 92.87 - 0.3333A - 2.05B + 0.0000AB + 0.0000A² + 0.6500B². Contour plots (Figure 4) illustrate DR@10h, while 3D response surface plots depict MS.
The contour and response surface plots illustrate that increasing AIFM and carbopol 934P levels enhances drug mucoadhesion time. The predicted R² value (0.8689) aligns well with the adjusted R² (0.9713), maintaining a difference below 0.2, confirming model reliability. Adequate Precision, measuring signal-to-noise ratio, is 20.169 (>4 indicating a strong signal).
ANOVA details of MS
The model's F-values confirm its significance, with minimal noise probability. Significant terms include A, B, and B², while values > 0.1 are insignificant. The predicted R² (0.9919) closely aligns with the adjusted R² (0.9981), maintaining a difference < 0.2, ensuring model reliability. Adequate Precision (75.368) exceeds the threshold of 4, indicating a strong signal. Contour and response surface plots show that increasing Carbopol 934P and AIFM levels enhances the MS of AAMM. The mathematical model MS = +18.57 + 0.3500A + 0.6667B + 0.1000AB - 0.1500A² - 0.2000B² allows for predicting outcomes based on factor levels.
Discussion
The DSC analysis showed a slight shift in the endother-mic peak (from 256.19ºC to 235.84ºC) when interacting with excipients, suggesting molecular interactions. However, these interactions did not compromise AVR’s stability, ensuring its functionality within the formulation. The FTIR spectra further supported this, as the core structure of AVR remained intact in the formulation, with key functional group peaks retained in the blend.
The QTPP approach facilitated a systematic identification of CQAs essential for developing AAMM formulations. This approach ensured the final product met predefined quality standards, and the QbD methodology provided a deeper understanding of how AIFM and Carbopol 934P influenced critical parameters, such as drug release and mucoadhesive strength. Combining these approaches allowed for a robust and reproducible preparation method, ensuring the formulation achieved desired therapeutic outcomes while maintaining consistent performance.
The formulations exhibited favorable flow and compressibility properties, with narrow ranges for Carr’s index values and Hausner ratios below 1.15, indicating minimal inter-particulate friction and excellent compressibility. The angle of repose values confirmed the ease of flow, critical for uniform die filling during compression. Formulations F-7 and F-8, in particular, demonstrated optimal flowability and compressibility, making them suitable candidates for further development and scale-up.
Particle size analysis revealed a relatively narrow size distribution in AAMM formulations, crucial for ensuring uniform drug release and mucoadhesion. The larger particle size in F-5 may be due to specific formulation parameters, such as excipient concentrations or process conditions. This variation may influence drug release kinetics and surface area, suggesting that F-5 warrants further investigation for its potential impact on dissolution and bioavailability.
High percentage yields across all formulations indicated an efficient, reproducible preparation method, with F-4 and F-5 yielding notably higher percentages. These results suggest optimized processing parameters and minimal product loss, making F-4 and F-5 promising candidates for large-scale manufacturing and potential commercialization due to their cost-effectiveness.
The EE values indicated effective encapsulation of AVR, with formulations F-6 and F-4 showing superior EE. This high EE suggests optimal polymer-drug interactions, critical for ensuring sustained drug release and therapeutic efficacy. The higher EE in these formulations makes them ideal candidates for further pharmacokinetic and stability studies.
Swelling behavior analysis demonstrated the significant role of AIFM in influencing mucoadhesion and drug release. Minimal swelling in formulations F-1 to F-3, with lower AIFM concentrations, limited water absorption, and matrix expansion, potentially affecting drug release. In contrast, higher swelling indices in F-7 to F-9, particularly F-9, were attributed to polar compounds in AIFM, which facilitate water absorption, leading to enhanced mucoadhesion and prolonged drug release. This suggests that formulations with higher AIFM concentrations are more suitable for sustained drug delivery applications.
The observed increase in mucoadhesion with higher AIFM content can be attributed to the enhanced interaction between the mucoadhesive polymer and mucosal surfaces. AIFM likely possesses greater adhesive properties, leading to stronger bonds with the mucus layer and improved mucoadhesive performance. This aligns with previous studies, which have shown that increasing the concentration of mucoadhesive agents generally enhances adhesion strength.
The rapid AVR discharge observed in F-1 and F-2 formulations suggests they can sustain drug release over an extended period, ensuring therapeutic concentrations are maintained. These formulations demonstrated efficient drug release, which is essential for controlled-release systems. However, exploring other formulations (F-3 to F-9) would be beneficial for further optimization of release profiles.
The observed linearity in the concentration range of 0-10 μg/ml confirms the method's precision, which ensures consistent measurement of AVR content and reproducibility across batches.
Fitting the discharge data to the Higuchi model indicated that AVR release from AAMM formulations is primarily diffusion-driven, a characteristic typical of matrix-based controlled-release systems. This release mechanism supports sustained drug delivery, ensuring consistent long-term therapeutic effects. The superior discharge profile of F-2 suggests it is particularly effective in maintaining a steady release rate, likely due to its optimized composition and excipient formulation.
The high F-value and P-values <0.05 confirm the model’s robustness and the significance of factors A, B and B² in influencing the mucoadhesion and dissolution properties of the formulations. These findings guide future optimization efforts by identifying key contributing factors that impact the final product’s performance.
The diagnostic plots for drug release and mucoadhesion strength confirm the model's reliability in predicting formulation performance. These findings suggest the optimization process, supported by statistical analysis and experimental data, is robust and suitable for achieving the desired formulation characteristics. The results provide valuable insights for future adjustments to enhance drug release and mucoadhesive strength, optimizing AAMM formulations for controlled drug delivery applications.
Conclusion
The study emphasizes the importance of mucoadhesive polymers in modulating the release rate and amount of Acyclovir (AVR), which improves the therapeutic efficacy of mucoadhesive drug delivery systems (MDDS). These polymers modulate AVR release, increasing the therapeutic efficiency of the system.
The gastric MDDS remains in the stomach for extended periods, promoting gradual AVR discharge. Nine formulations combining AIFM and Carbopol 934P were developed, with Formulations F-1 and F-2 showing significant AVR release by the 10th hour, outperforming other formulations. All formulations demonstrated increased mucoadhesive strength with higher AIFM content.
Acknowledgments
The authors are thankful to the management of R R College of Pharmacy, Bengaluru for the encouragement and support.
Conflict of Interest
The authors declare no conflict of interest.
Supporting File
References
1. Ahad HA, Haranath C, Rahul Raghav D, et al. Overview on recent optimization techniques in gastro retentive microcapsules by factorial design. Int J Pharm Sci Res 2019;10(9):247-254.
2. Babu GN, Menaka M, Ahad HA. Neem Fruit Mucilage-Aided Mucoadhesive Microspheres of Acyclovir Using 32 Factorial Design With Design- Expert Software 2022;24(1):17-27.
3. Babu GN, Muthukarupan M, Ahad HA, et al. Fabrication and Preliminary Assessment of Neem Fruit Mucilage as Mucoadhesive Abetting Assets with Methpol-934P for Acyclovir Delivery from Mucoadhesive Microcapsules. Biomed and Phar-macol J 2022;15(4):2179-2184.
4. Babu GN, Muthukarupan M, Ahad HA. Neem Fruit Mucilage Impact on Acyclovir Release at Different Intervals: A Central Composite Design Screening. International Journal of Pharmaceutical Research & Allied Sciences 2021;10(4):131-141.
5. Ahad HA, Chinthaginjala H, Priyanka MS, et al. Datura stramonium Leaves Mucilage Aided Buccoadhesive Films of Aceclofenac using 3² Factorial Design with Design-Expert Software. Indian Journal of Pharmaceutical Education & Research 2021;55(4):s396-s404.
6. Shobana N, Prakash P, Samrot AV, et al. Purification and characterization of gum-derived polysaccha-rides of Moringa oleifera and Azadirachta indica and their applications as plant stimulants and bio-pesticidal agents. Molecules 2022;27(12):3720.
7. Yadiki MN, Suggala VS, Puchalapalli DSR, Ahad HA. Temperature and Exposure Time Impact on the Extraction of Opuntia ficus-indica and Opuntia dillenii Cladodes on % Yield as a Response: Screening using Design Expert Software. Global Journal of Medical, Pharmaceutical, and Biomedical Update 2022;17:17.
8. Shravani Y, Ahad HA, Haranath C, et al. Past Decade Work Done On Cubosomes Using Factorial Design: A Fast Track Information for Researchers. Int J Life Sci Pharma Res 2022;11(1):P124-135.
9. Mundarinti SHB, Ahad HA. Impact of Pistacia lentiscus Plant Gum on Particle Size and Swelling Index in Central Composite Designed Amoxycillin Trihydrate Mucoadhesive Microspheres. Ind J Pharm Edu Res 2023;57(3):763-772.
10. Hossain MS, Jahan S, Rahman SAR, et al. Design expert software assisted development and evaluation of empagliflozin and sitagliptin combination tablets with improved in-vivo anti-diabetic activities. Heliyon 2023;9(3):e14259.
11. Noreen S, Hasan S, Ghumman SA, et al. Formulation, Statistical Optimization, and In Vivo Pharmacodynamics of Cydonia oblonga Mucilage/Alginate Mucoadhesive Microspheres for the Delivery of Metformin HCl. ACS omega 2023;8(6):5925-5938.
12. Harsha SS, Ahad HA, Haranath C, et al. Exfoliation Technique of Composing and Depictions of Clopidogrel Bisulphate Afloat Microspheres. J Evol Med Dent Sci 2020;9(14):1156-1161.
13. Perkušić M, Nodilo LN, Ugrina I, et al. Tailoring functional spray-dried powder platform for efficient donepezil nose-to-brain delivery. Int J Pharm 2022;624:122038.
14. Kashani-Asadi-Jafari F, Aftab A, Ghaemmaghami S. A machine learning framework for predicting entrapment efficiency in niosomal particles. Int J Pharm 2022;627:122203.
15. Bouassida M, Manigniavy SA, Azaiez D, et al. New approach for characterization and mitigation of the swelling phenomenon. Frontiers in Built Environment 2022;8:836277.
16. Dawre S, Waghela S, Saraogi G. Statistically designed vitamin D3 Encapsulated PLGA microspheres dispersed in thermoresponsive in-situ gel for nasal delivery. J Drug Deliv Sci Technol 2022;75:103688.
17. Dandagi P, Kerur S, Mastiholimath V, et al. Polymeric ocular nanosuspension for controlled release of acyclovir: in vitro release and ocular distribution. Iran J of Pharm Res 2009;8(2):79-86.